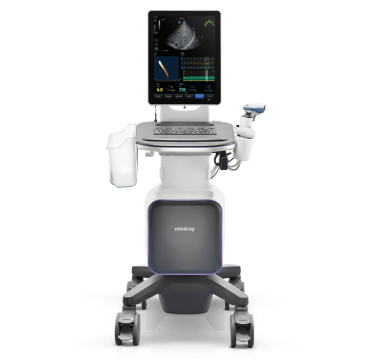
Mindray’s total vertical integration allows us to better control every element of our ultrasound systems, enabling better innovation, quality, and cost.
A System Changing
As home to one of the costliest healthcare systems in the world, the U.S. is one of many countries globally fighting to find a constructive way forward with the shift to value-based care. According to the Institute for Healthcare Improvement, “countries with health systems that
out-perform the U.S. are also under pressure to derive greater value for the resources devoted to their healthcare systems1 .” Aging populations paired with increasing numbers of comorbidities and chronic health now present a global challenge, putting new demands on social and medical services. As the industry continues this shift, hospitals and healthcare providers continue to seek out new and improved ways to enhance the level of care patients receive, and realize the triple aim value approach. This increase in value may look like preventative rather than reactive care, more comprehensive public health education, improved quality of care through improved resources, and/or lower costs for hospitals that can ultimately be passed to their patients. At Mindray, our role in the switch to value-based care is clear: bringing you the highest quality products in the industry, at an accessible price point. How do we do this? By controlling the elements.
Total Vertical Integration
A key advantage in Mindray’s value proposition is the wholly integrated product development process from research and innovation, through design, manufacturing, to final testing and validation, as well as a closely controlled procurement process. Mindray has adopted a robust, vertically integrated production model, to better control quality and reduce risks, as well as improve overall efficiency.
Before a product receives Mindray’s stamp of approval, a number of rigorous tests must be carried out including:
- Individual component testing
- Functional testing
- Durability testing
- EMC
- Hazardous voltage
- Radiation
- Salt-spray corrosion
- Waterproofing
- Shock and vibration resistance testing
- Extreme temperature exposure tests
- Final quality control (FQC) and outgoing quality control (OQC)
In addition, each day a statistical process control collects production parameters and analyzes any abnormalities so that the quality and reliability of each product can be guaranteed.
How it’s Done
Whether it’s the screws that hold your system together or the touchscreens incorporated to enhance your workflow, Mindray maintains complete control over the production of its ultrasound systems from ideation to build. We visualize every step of the manufacturing process through a set of intelligent management measures. By working in a vertically integrated system, Mindray can maintain complete quality control and ensure stringent criteria and traceability throughout the entire process. To ensure that Mindray products are safe, effective, and reliable, we have identified five key phases to carry out strict quality control measures; namely engineering design, supplier quality assurance, manufacturing, service and training, and post-market surveillance. The work of each link in this cycle is carefully managed due to its impact on the next one and eventual effect on the quality of the product provided to you, our user. After all of these stringent guidelines are met, we are able to efficiently and effectively transport high quality medical equipment to every corner of the world on a daily basis.
Now Let’s Change the Future
Quality is the lifeblood of Mindray and our products, because we understand the vital role of high-quality medical devices in patient treatment. Through higher quality, lower cost solutions like Mindray Ultrasound, you and your organization can continue tackling the industry’s shift with confidence. Regardless of how we get there, we are all on the same journey, racing towards the same, ever-moving finish line, that looks a lot like better healthcare for all.